Crude Oil Refining Technology
In technical collaboration with
Erpek Engineering & Consulting, Istanbul, Turkey
Separation
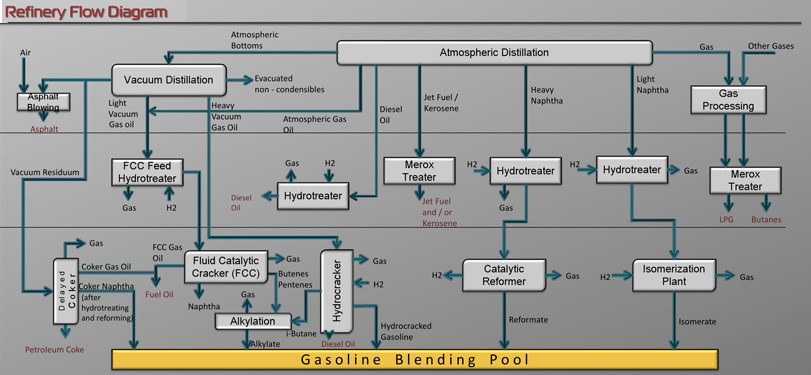
Modern separation involves piping crude oil through hot furnaces. The resulting liquids and vapors are passed into distillation tower so that hydrocarbons can be separated via their boiling point. In the first step separation tower the lightest fractions, including gasoline and liquid petroleum gas (LPG), vaporize and rise to the top of the tower, where they condense back to liquids. Medium weight liquids, including kerosene and diesel oil distillates, stay in the middle. Heavier liquids, called gas oils, separate lower down, while the heaviest fractions with the highest boiling points settle at the bottom.
In the secondary separation processing unit consists of vacuum distillation column. Vacuum distillation is a part of the refining process that helps to produce petroleum products out of the heavier oils left over from atmospheric distillation. Vacuum distillation produces several types of gas oil. These are slightly heavier than middle distillates such as jet fuel, kerosene, and diesel.
Treatment
Treatment is the final process of refining, and includes combining processed products to create various octane levels, vapor pressure properties, and special properties for products used in extreme environments. One common example of treatment is the removal of sulfur from diesel fuel, which is necessary for it to meet clean air guidelines. Treatment is highly technical and is the most time consuming step of refining.
Another treatment type of petroleum refining is merox treatment which is an extractive catalytic chemical process removing trace sulfidic components, mercaptans, from LPG, propane, butanes, light naphthas. Kerosene and jet fuel by converting them to liquid hydrocarbon disulfides.
Conversion
Cracking and rearranging molecules adds value to the products. This is where refining's fanciest footwork takes place-where fractions from the distillation towers are transformed into streams that eventually become finished products. The process is simply changing one kind of hydrocarbon into another. The most widely used conversion method is called cracking because it uses heat and pressure to "crack" heavy hydrocarbon molecules into lighter ones. A cracking unit consists of one or more tall, thick-walled, bullet-shaped reactors and a network of furnaces, heat exchangers and other vessels. Cracking and coking are not the only forms of conversion. Other refinery processes, instead of splitting molecules, rearrange them to add value. Alkylation's, for example, makes gasoline components by combining some of the gaseous byproducts of cracking. The process, which essentially is cracking in reverse, takes place in a series of large. Horizontal vessels and tall. skinny towers that loom above other refinery structures. Reforming uses heat moderate pressure and catalysts to turn naphtha, a light, relatively low-value fraction. into high-octane gasoline components.